The U.S. Food and Drug Administration (FDA) has determined that short-haired cattle produced through CRISPR gene-editing technology are safe for human consumption. The cattle, known as PRLR-SLICK, were the first to receive an FDA "low-risk determination for enforcement discretion" after the agency determined the intentional genomic alteration (IGA) of the two genome-edited cattle does not raise any safety concerns.
Produced by Acciligen with climate change in mind, the cows have a genetic trait that gives them a short, sleek coat which is said to help the animals cope with hot weather more effectively. The FDA's low-risk determination means the agency does not expect Acciligen, a "precision breeding" company, to seek regulatory approval before marketing products from the cattle.
The FDA spent years reviewing the two other genetically altered animals approved for human consumption—a faster-growing salmon and a pig the agency determined was safe for consumption by people with meat allergies. However, the review process for the CRISPR beef cattle took less than a year because the FDA noted the gene-editing results in the same slick-hair trait seen in cattle that are found in conventional agriculture. Talking about the Mar. 7, 2022 approval, Steven Solomon, director of the FDA Center for Veterinary Medicine, said:
"We expect that our decision will encourage other developers to bring animal biotechnology products forward for the FDA's risk determination in this rapidly developing field, paving the way for animals containing low-risk IGAs (intentional genomic alterations) to more efficiently reach the marketplace."
Looking closer at Acceligen, the company website says that most of its workers have backgrounds in the farm industry. The company explains that "precision breeding" is different from conventional breeding or genetically modified organisms (GMO) in that it allows a "highly desired trait" that may typically take years to show up to be expressed in "just one breeding cycle."
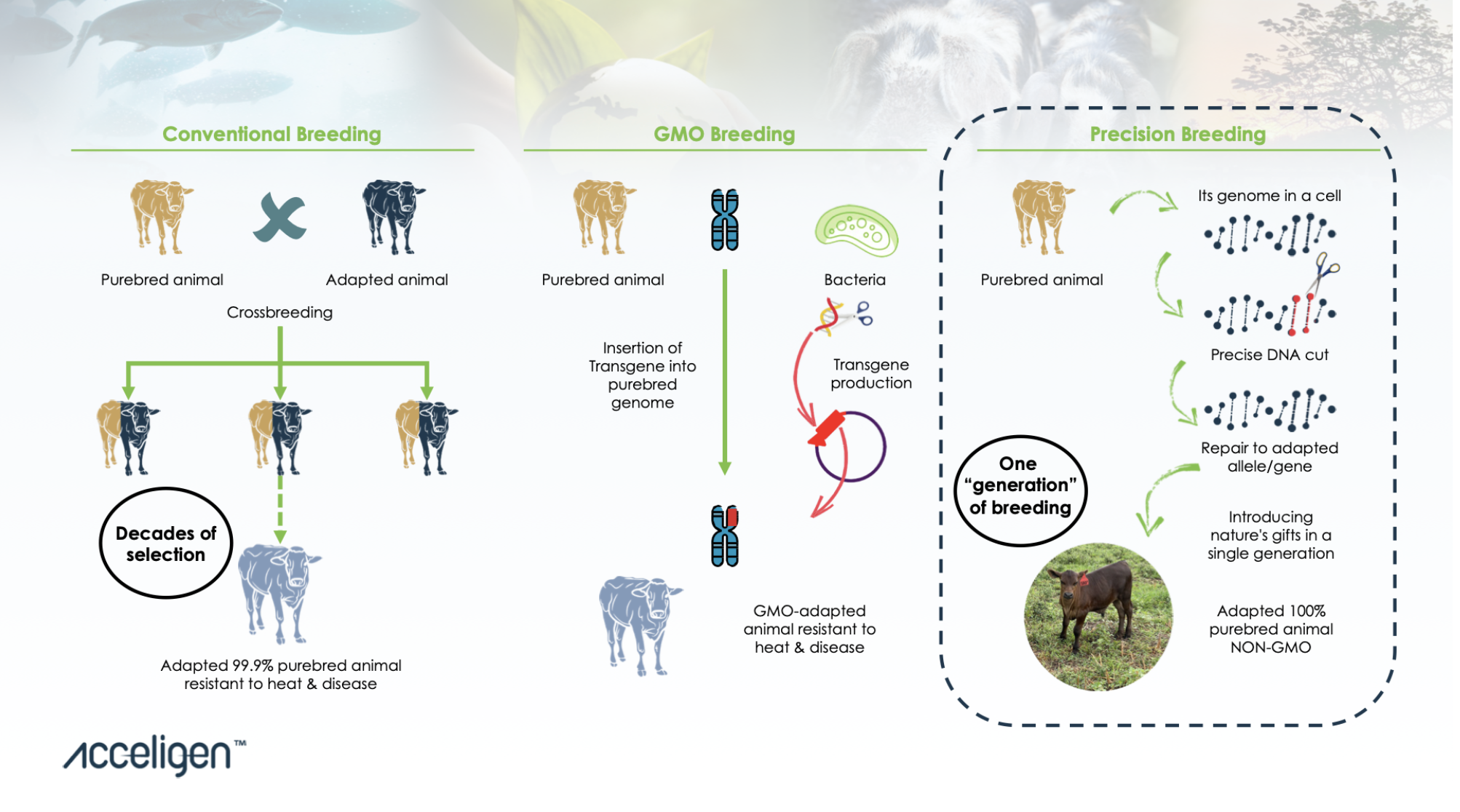 Screenshot / Acceligen
Screenshot / Acceligen
In 2020, Acceligen, a Recombinetics Inc. company, announced it received a $3.68 million grant from the Bill & Melinda Gates Foundation to develop bovine genetics optimized with characteristics desirable to smallholder dairy farmers in the Sub-Saharan Africa (SSA) dairy systems. The breeding program will deploy a "suite of traits" from its discovery pipeline into commercially important dairy animals "with high genetic merit for production and durability." Tad Sonstegard, CEO of Acceligen and project lead for the grant, said of the endeavor:
"When we combine gene editing with top merit animals using advanced reproductive technologies from our partners Kheiron (Pilar, Argentina) and TransOva Genetics (Sioux Center, Iowa), we can make significant genetic improvement for well-adapted, high yielding dairy cows. Our goal is to get these animals into the hands of smallholder farmers."
K25Yvq8NV_oW-h4hG7tslQ
Researchers have found numerous applications for CRISPR in animals, such as creating disease-resistant chickens and pigs and mosquitos unable to bite or lay eggs. They've also got many projects underway, like producing disease-resistant crops—including grapes for winemaking. Furthermore, they're working to genetically alter pigs so their organs can be transplanted into humans. Also, by tweaking the genomes of certain birds, a project is underway to bring extinct species such as the passenger pigeon back to life.
In a noteworthy landmark operation earlier this year, a 57-year-old man was near death when, in January, he received a genetically edited pig heart—the first between species transplant that had been "hailed as a success" by xenotransplantation—the process of moving tissues between species. However, nearly 40 days after the transplant, the man took a turn for the worse, and two months later, he passed away. Initially, the hospital noted "no obvious cause identified at the time of his death." Nevertheless, MIT Technology Review reported on May 4 that the patient's heart was infected with a pig virus. Describing the complicated process, MIT wrote:
The biggest obstacle to animal-organ transplants is the human immune system, which ferociously attacks foreign cells in a process called rejection. To avoid rejection, companies have been engineering pigs—removing some genes and adding others—to give their tissue a stealth profile that hides from immune attack.
Transferring pig viruses to humans has been a worry—some fear xenotransplantation could set off a pandemic if a virus were to adapt inside a patient's body and then spread to doctors and nurses. The concern could be serious enough to require lifelong monitoring for patients.
 Screenshot / FDA
Screenshot / FDA
Many researchers expect CRISPR-based therapies (which received significant funding from Bill Gates and others several years ago) may someday be used to cure hereditary diseases. Scientific America (SA) points out that in 2020, U.S. researchers began the first clinical trials injecting CRISPR straight into living humans, aiming to repair a genetic mutation that causes blindness.
Indeed, while CRISPR "reliably cuts DNA where we want it to," recent experiments have shown it can also affect genes far off-target. As noted by SA, even if we could get it to work reliably, many experts have flagged ethical worries about using the technology for eugenics and "designer babies." SA wrote, "If parents can one day pay scientists to edit their babies' DNA, making them stronger and smarter, CRISPR could make the world even more unequal and prejudiced." SA contends that given the dangers of editing the human genome, researchers are still a ways off from the widespread use of CRISPR in medicine, adding:
"CRISPR has given science a tool to reliably tinker with the code of life. But the question remains: can we do so safely and ethically while avoiding the unintended consequences of such power?"
There is no definite word yet on when consumers might see beef from CRISPR cows in US grocery stores, but it could be as soon as two years. It is also unclear whether meat from the gene-edited cows will be labeled differently from conventional beef.


