On Monday it was announced that a Covid-19 vaccine can prevent nine out of ten people from getting the virus. The developers, Pfizer and BioNTech, said it had been tested on 43,538 people (42 percent had diverse backgrounds) no serious safety concerns were observed by an external data monitoring committee. Safety and additional efficacy data continue to be collected, and the intention is to submit the vaccine for Emergency Use Authorization (EUA) to the U.S. Food and Drug Administration (FDA) in the third week of November.
What is the new vaccine and how effective is it?
The vaccine works by teaching the immune system to fight Covid-19. Unlike vaccines we are used to, it is an RNA (Ribonucleic acid/RNA is a polymeric molecule essential in various biological roles) vaccine that relies on a little fragment of the virus' genetic code. This fragment starts to make part of the virus inside the body, the immune system recognizes this is foreign and attacks it. The vaccine would be taken in two doses—three weeks apart.
Has this type of vaccine ever been used before?
This will be the first RNA vaccine to be approved for use in humans. It is not a new concept, though, having been researched and used in prior clinical trials for other diseases. Various regulatory agencies around the world will need to approve it for use in their countries.
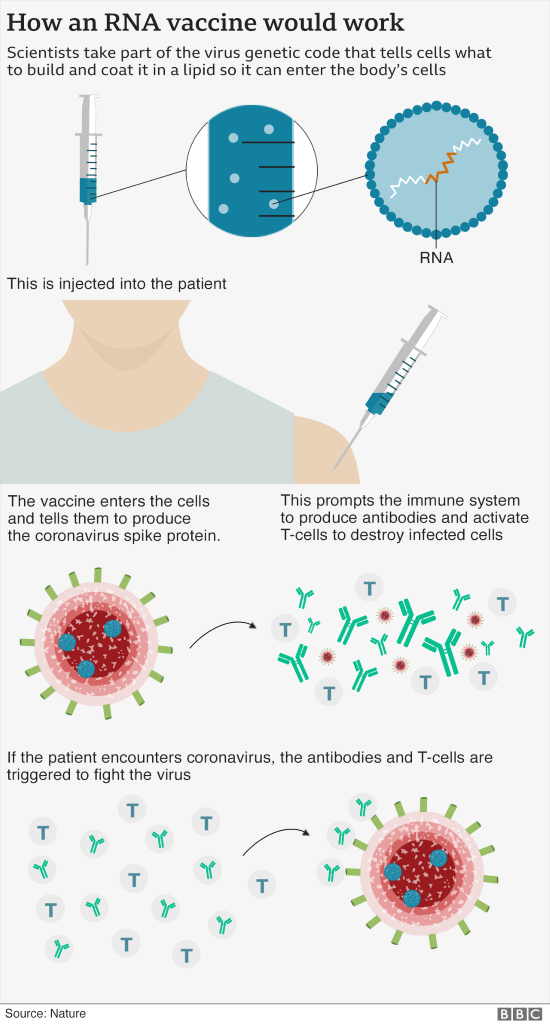
Researchers had expected new coronavirus vaccines to be about 60 to 70 percent effective, as even a good flu vaccine only protects within the 40-60 percent range. Ugur Sahin, CEO of BioNTech, says a 90 percent rate of effectiveness is “extraordinary, and indicates a way Covid-19 can be controlled.” Pfizer and BioNTech will produce 50 million doses of the vaccine by the end of 2020, another 1.3 billion by the end of 2021.
Logistical challenges to overcome
The vaccine has to be kept at minus 80c while moving from the manufacturing lab to vaccination distribution venues. The jab then needs to be thawed before administering it to a patient and can be stored in a normal fridge for a few days before being given.
Scientists are not sure whether its protection will last, or if it will be needed annually like a flu jab. Data also does not yet show whether it protects all age groups equally.
What if Covid-19 mutates?
It is natural for viruses to mutate; it is how they survive. The problem will be if they mutate in a way that changes their behavior. Presently, scientists have not seen that, but the virus has the ability to change and make vaccines less effective. This is in fact what happens each year when a new flu vaccine is developed to try to match the strains of flu going around.
Scientists are confident that RNA vaccine technology is very easy to tweak and this will not be a major problem. Additionally, Pfizer’s vaccine went from drawing board to highly effective outcomes in the fastest time ever, so testing will be ongoing. Scientists globally working on the effort through, Operation Warp Speed, may explain the shorter time frame.
Media gives no credit to the Trump administration?
Following Pfizer’s announcement, reporting by legacy media outlets including the New York Times, Washington Post and Newsweek, sought to distance the Trump administration from this development. The New York Times quoted the head of vaccine development at Pfizer, Kathrin Jansen, as saying, “We have never taken money from the U.S. government or from anyone.” The NYT then claimed Pfizer was never a part of Operation Warp Speed.
This was not actually the case. Pfizer’s press release from July stated that the U.S. government placed an initial order of 100 million doses for $1.95 billion, as well as money to aid distribution through the Trump Administration’s Operation Warp Speed.
It is correct that no money went specifically into the vaccine’s research and development and Pfizer later revised Jansen’s statement: “This page has been updated to clarify Jansen’s comments following further details from a Pfizer spokesperson. The spokesperson clarified that its vaccine is linked to Operation Warp Speed, though its research and development has not taken funding from the federal government. The headline was also updated for clarity.”
Did Biden know first?
President Trump was pleased to report news that a vaccine would be coming soon at 12.30 p.m. on Monday, Nov. 9, but commentator Mark Levin asked whether Biden was made aware of this first.
Levin said on his radio show that White House aides had relayed that they and the President found out on Monday, but Biden said he found out about it on Sunday night. He said, his "public health advisors," told him but that does link up with Pfizer’s own announcement that they received results of their first interim external efficacy analysis on Sunday.

Pfizer COVID vaccine not created with fetal cells
Pro-lifers and those with strong religious beliefs strongly oppose vaccines developed using fetal cells taken from aborted fetuses. A check of Pfizer’s vaccine development process, revealed that it came about through genetic sequencing on computers and that no fetal cells were used. This double-checks out with the pro-life Lozier Institute which has listed the vaccine as “ethically uncontroversial.”
Carol King received a first-class BA (honors) in History and Politics from Stirling University, along with an exceptional commendation for a study on U.S. public opinion and Foreign Policy. She also completed a year of study at the University of London before taking up a Graduate Proctor Fellowship at Princeton University. She further completed an MPhil in American Politics at Dundee University. Aspiring to be a writer/commentator on American politics, she now writes for UncoverDC.
Twitter: @CarolKing561


